|
group 1 to group 17 elements tend to attain the noble gas configuration and this is known as the octet rule. Octet rule/Octet fulfillment: Let us look at the periodic table. Here comes the concept of octet fulfillment. 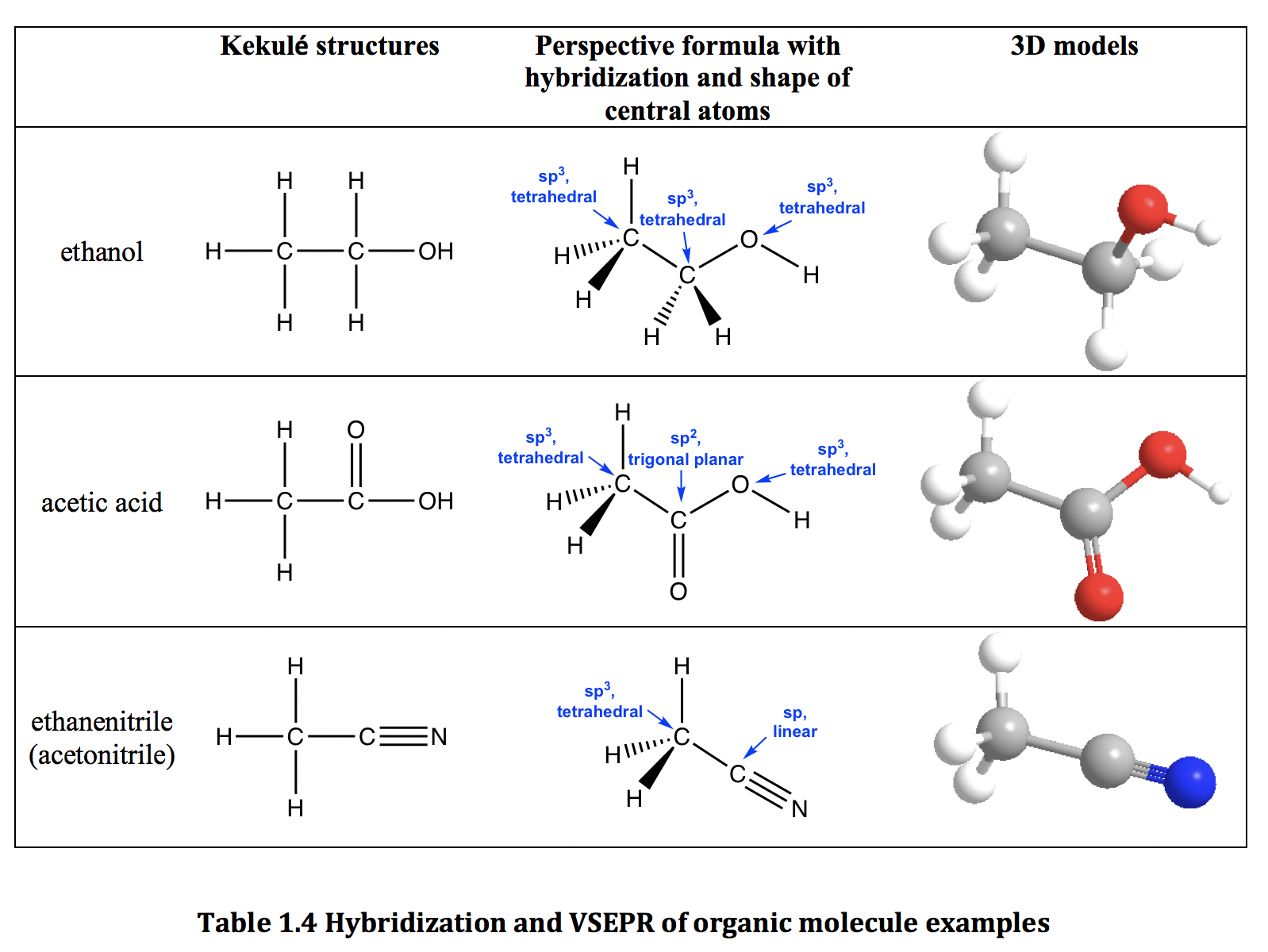
We have put all the 10 valence electrons surrounding the constituent atoms of the CN ionic molecule. We will henceforth place the electron dot notations. We have placed both the carbon and nitrogen atoms as atomic symbols here. Now, we will proceed to draw a simple sketch or skeletal diagram of cyanide ion. Since CN is a diatomic ion, there is no concept of a central atom here. total number of valence electrons = 4 + 5 + 1 = 10. Nitrogen belongs to group 5/15 and has a valency of 5.Īlso, we need to take into account the electron that provides the negative charge to the CN- ion. Finding out the Lewis Structure of CN ionĪ cyanide anion consists of carbon and a nitrogen atom.Ĭarbon belongs to group 4 or 14 of the periodic table and therefore has 4 valence electrons. Here, the valence electrons are denoted as dots and we use straight lines to denote the type of bond formed between the valence electrons.Īlthough Lewis Structure does not give us many details about the bonding, it is the initial step towards understanding the 3D molecular shape or finding out the nature of hybridization. Lewis Structure is a simplified and easy diagrammatic representation of the internal structure of a chemical compound. The outermost shell is known as the valence shell that determines the valency i.e the combining capacity of the atom with other atoms for molecule formation. Here since an oxygen atom has an atomic number of 8, we have six electrons in the outermost shell. If we have a look into the above-mentioned diagram, we can see that it is a sketch of an oxygen atom. Let us now discuss the chemical bonding inside a cyanide ion to understand its chemical and physical properties in a better manner.Ĭonclusion CN Lewis Structure What is Lewis Structure? It can cause asthma, bronchitis, acidosis, prenatal injuries, and even brain edema. Reaction with water can lead to fumes and fire generation as well. Apart from this, cyanides have applications as food additives and in jewelry-making industries.Ĭyanide salts are however toxic and poisonous for human bodies. It can act as a reducing agent.Īs HCN, we find use in gas chamber executions and preparation of pesticides and insecticides.Īs NCN, it is used for gold and silver mining. We can find CN- ion in both organic molecules like acetonitrile and inorganic molecules like potassium cyanide.Ĭhemical compounds containing CN- ion have a lot of applications in the present-day world. It carries a charge of -1 and is a conjugate base of hydrogen cyanide (HCN).Ĭyanides are released in nature via cyanogenic compounds and also produced by several microorganisms. It belongs to the cyano group and consists of carbon and a nitrogen atom having a triple bond. All that was known in the early days was that every purified sample of C 2H 5Br, no matter how prepared, had a boiling point of 38 oC and density of 1.460 gml −1.CN is known as cyanide which exists as a pseudohalide anion. When looking at the possible structures of the compound C 2H 5Br there are several possible structural formulas.here was a serious problem as to whether these formulas represent the same or different compounds. But how did they know? A question came up when looking at ethane with a bromine substituent (C 2H 5Br). The tetrahedral geometry of carbon was predicted as far back as 1874. In Section 3.7 we will learn more about the implications of rotational freedom in sigma bonds, when we discuss the ‘conformation’ of organic molecules This means, in the case of ethane molecule, that the two methyl (CH 3) groups can be pictured as two wheels on a hub, each one able to rotate freely with respect to the other. Because they are formed from the end-on-end overlap of two orbitals, sigma bonds are free to rotate. The orientation of the two CH 3 groups is not fixed relative to each other. The C-C-H bond angles in ethane are 111.2 o which is close to the what is expected for tetrahedral molecules. The carbon-hydrogen σ bonds are slightly weaker, 421 kJ/mol, than those of methane. 
The σ carbon-carbon bond has a bond length of 154 pm, and a bond strength of 377 kJ/mol. 
Each of the remaining sp 3 hybrid orbitals overlaps with the s orbital of a hydrogen atom to form carbon–hydrogen σ bonds. This orbital overlap is often described using the notation: sp 3(C)- sp 3(C). 
An sp 3 orbital of one carbon atom overlaps end to end with an sp 3 orbital of the second carbon atom to form a carbon-carbon σ bond. In ethane (CH 3CH 3), both carbons are sp 3-hybridized, meaning that both have four bonds with tetrahedral geometry. The simplest molecule with a carbon-carbon bond is ethane, C 2H 6.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
